Easy to LIVE
FOR DOGS

Easy to LIVE
FOR DOGS
DID YOU KNOW?
Three tablet strengths with a narrow weight range for each, means it's easy to be accurate about dosing Cimalgex® and get the pain relief just right.
DID YOU KNOW?
Three tablet strengths with a narrow weight range for each, means it's easy to be accurate about dosing Cimalgex® and get the pain relief just right.

Bring back their smile...

Bring back their smile...
Make it easier to live with better pain management
Easy snap, multi-scored tablets to match narrow weight brackets, without under-dosing.
Proven efficacy for long and short-term pain management.1,4,5
Rapidly available in blood for effective pain reduction.1
Proven tolerance for long term use.9-12
Accurate, effective dosing for more comfortable dogs, quickly

Maximum plasma level in 2 hours 15 minutes after giving tablets.1

Proven efficacy in short and long-term pain management1,4,5
Studies show Cimalgex® provides substantial improvement in clinical signs of canine OA (pain, locomotion, lameness), as judged by vets and pet owners.
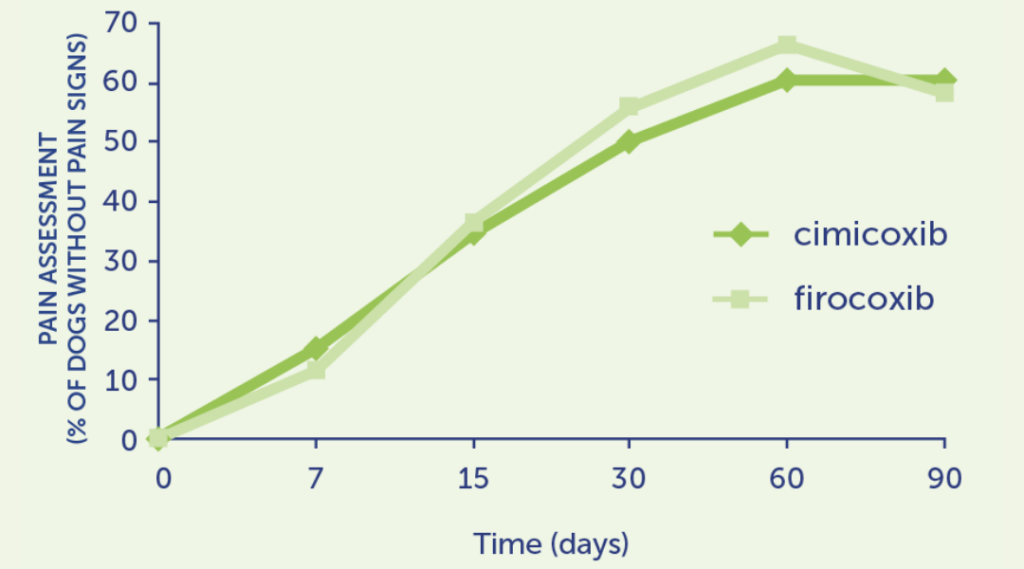
EVIDENCE Effective in chronic analgesia5
Osteoarthritis Field Trial
Assessment of pain in dogs treated with Cimalgex® for 90 days.


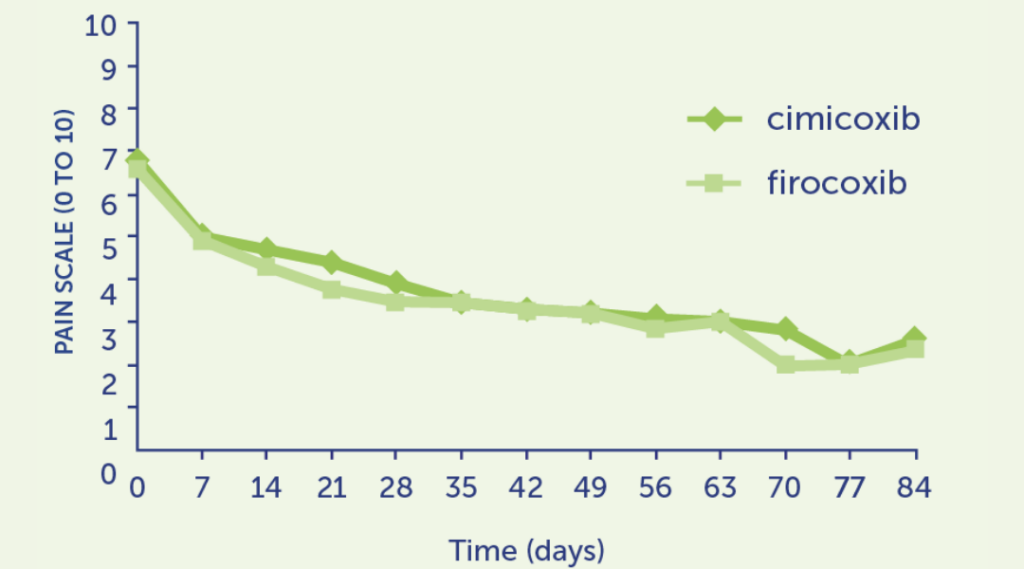


EVIDENCE Effective in acute analgesia4
Peri-operative Pain Field Trial
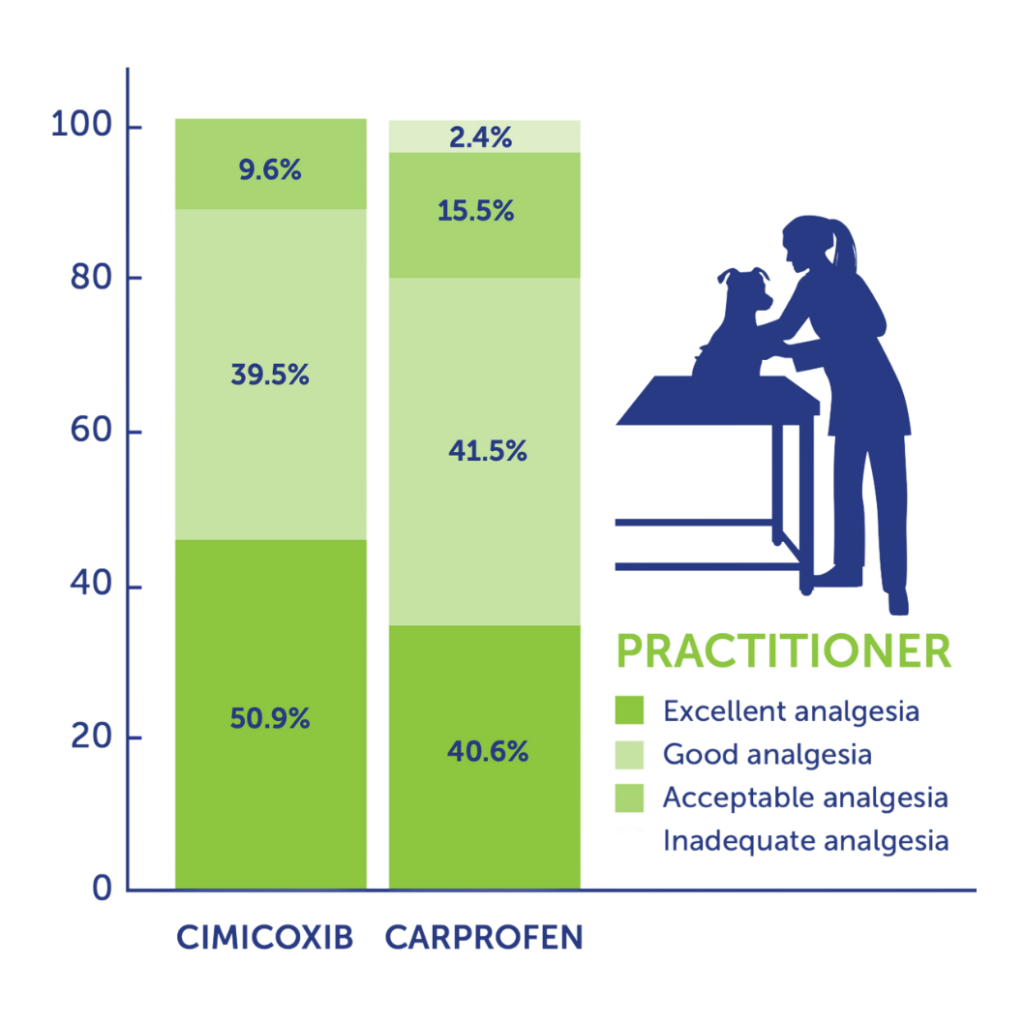
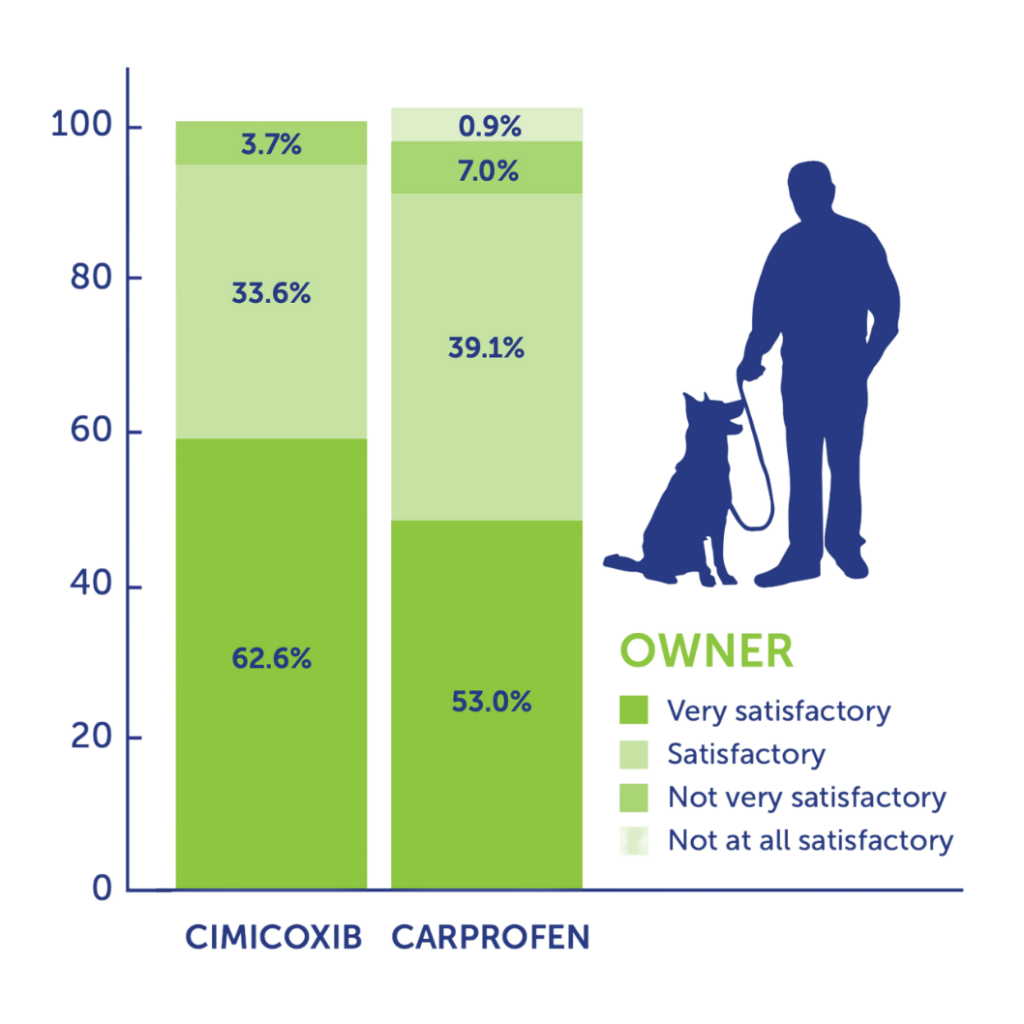
More than 90% of vets and pet owners judged Cimalgex® to have good to excellent analgesic potential for controlling post operative pain (orthopaedic or soft tissue).
There were no reports of inadequate analgesia in the Cimalgex® treated groups.

TOP TIP

Think slow, Act fast
Take time to consider your decisions and the evidence, so that when the time comes you can respond with a fast acting solution.
Proven tolerance for long term use5,9,11,12
Tolerance trials
QUICK REFERENCE
●
At recommended dose levels over 26 weeks, no GI signs on endoscopy.9
●
Cimicoxib is eliminated mainly in the faeces.10
●
At 5 x the recommended dose for 39 weeks, no changes to BUN or creatinine.11
●
At 6 x the recommended dose, across 5 field trials, no changes to hepatic enzymes.4,5,9,12
EVIDENCE No active cimicoxib is eliminated in the urine.10

Elimination
Faeces
Urine
Cimicoxib
15%
0%
Inactive metabolite
62%
14%
EVIDENCE At 6 x recommended doses, across 5 field trials, no changes to hepatic enzymes.4,5,9,11,12
In five experimental and field clinical trials, no statistical alterations of hepatic enzymes were observed, even in dogs treated with up to 6 times the licensed dose.

Hepatic parameters (AST, ALT, ALP:U/L)
No statistical modifications observed

EVIDENCE In a 39 week tolerance trial of Cimicoxib given to healthy dogs at up to 5 times the recommended dose:11
No significant changes in blood urea nitrogen and creatinine levels were noted during the study.
EVIDENCE Trial: At recommended dose levels over 26 weeks, no GI signs on endoscopy9
A 26-week tolerance trial of 32 dogs across four treatment groups (placebo, 2 mg/kg/day, 6 mg/kg/day or 10 mg/kg/day) showed:
No treatment related findings (such as erosions, ulcerations or perforations) in the stomach and duodenum on digestive endoscopy, at all tested dose levels.
No statistical differences in the gastric and gastro-duodenal scores between the control group and treated animals.
A dose effect relationship was indicated for other gastro-intestinal effects. However, in the recommended dose (2mg/kg/day) group no gastro-intestinal differences were noted from the control group.
Body weight and food or water consumption were not affected by treatment with Cimalgex® at any dose level.
Always monitor potentially complicated cases as advised in the Cimalgex® SPC.3

* Scores for stomach lesions were ranked from score 0 (normal mucosa) to score 16 (diagnosed ulceration).
** Scores for duodenal lesions were ranked from score 0 (normal mucosa) to score 4 (diagnosed ulceration).
*** One score missing for hidden gastric mucosa (food ingestions) at control.
